 Global Information
Global InformationAtomoxetine information
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Strattera, others |
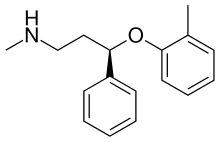
| Other names | (R)-N-Methyl-3-phenyl-3-(o-tolyloxy)propan-1-amine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603013 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Selective norepinephrine reuptake inhibitor |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 63 to 94%[7][8][9] |
| Protein binding | 98%[7][8][9] |
| Metabolism | Liver, via CYP2D6[7][8][9] |
| Elimination half-life | 4.5–25 hours[7][8][9][10][11] |
| Excretion | Kidney (80%) and faecal (17%)[7][8][9] |
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID |
|
| IUPHAR/BPS |
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEBI |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) |
|
| ECHA InfoCard | 100.120.306 |
| Chemical and physical data | |
| Formula | C17H21NO |
| Molar mass | 255.361 g·mol−1 |
| 3D model (JSmol) |
|
SMILES
| |
InChI
| |
| | |
Atomoxetine, sold under the brand name Strattera, is a medication used to treat attention deficit hyperactivity disorder (ADHD)[12] and, to a lesser extent, cognitive disengagement syndrome.[13][14][15] It may be used alone or along with psychostimulants.[16][17] It is also used as a cognitive and executive functioning enhancer to improve self-motivation, persistence, attention, inhibition, and working memory.[18][19][20] Use of atomoxetine is only recommended for those who are at least six years old.[12] It is taken orally.[12] Atomoxetine is a selective norepinephrine reuptake inhibitor and is believed to work by increasing norepinephrine and dopamine levels in the brain.[12][10] The effectiveness of atomoxetine is comparable to the commonly prescribed stimulant medication methylphenidate.[21][22][23][24]
Common side effects of atomoxetine include abdominal pain, loss of appetite, nausea, feeling tired, and dizziness.[12] Serious side effects may include angioedema, liver problems, stroke, psychosis, heart problems, suicide, and aggression.[12][25] There is a lack of data regarding its safety during pregnancy; as of 2019, its safety during pregnancy and for use during breastfeeding is not certain.[26][27]
It was approved for medical use in the United States in 2002.[12] In 2021, it was the 219th most commonly prescribed medication in the United States, with more than 1.9 million prescriptions.[28][29]
- ^ "Atomoxetine (Strattera) Use During Pregnancy". Drugs.com. 22 August 2019. Archived from the original on 22 March 2019. Retrieved 7 February 2020.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ Anvisa (15 September 2023). "RDC Nº 816 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 816 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 18 September 2023). Archived from the original on 19 October 2023. Retrieved 19 October 2023.
- ^ "Strattera 10mg hard capsules - Summary of Product Characteristics (SmPC)". (emc). 8 February 2021. Archived from the original on 8 October 2021. Retrieved 11 June 2022.
- ^ Cite error: The named reference
Strattera FDA labelwas invoked but never defined (see the help page). - ^ "Active substance(s): atomoxetine" (PDF). List of nationally authorised medicinal products. European Medicines Agency. 2016. Archived (PDF) from the original on 12 June 2022. Retrieved 12 June 2022.
- ^ a b c d e "Atomoxetine (Rx) – Strattera". Medscape Reference. WebMD. Archived from the original on 10 November 2013. Retrieved 10 November 2013.
- ^ a b c d e Cite error: The named reference
TGAwas invoked but never defined (see the help page). - ^ a b c d e "Atomoxetine Hydrochloride capsule [Mylan Pharmaceuticals Inc.]". DailyMed. Mylan Pharmaceuticals Inc. October 2011. Archived from the original on 10 November 2013. Retrieved 10 November 2013.
- ^ a b Sauer JM, Ring BJ, Witcher JW (2005). "Clinical pharmacokinetics of atomoxetine". Clinical Pharmacokinetics. 44 (6): 571–590. doi:10.2165/00003088-200544060-00002. PMID 15910008. S2CID 25708096.
- ^ Cite error: The named reference
PMID26314574was invoked but never defined (see the help page). - ^ a b c d e f g "Atomoxetine Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 4 April 2019. Retrieved 22 March 2019.
- ^ McBurnett K, Clemow D, Williams D, Villodas M, Wietecha L, Barkley R (February 2017). "Atomoxetine-Related Change in Sluggish Cognitive Tempo Is Partially Independent of Change in Attention-Deficit/Hyperactivity Disorder Inattentive Symptoms". Journal of Child and Adolescent Psychopharmacology. 27 (1): 38–42. doi:10.1089/cap.2016.0115. PMID 27845858.
- ^ Becker SP, Willcutt EG, Leopold DR, Fredrick JW, Smith ZR, Jacobson LA, et al. (June 2023). "Report of a Work Group on Sluggish Cognitive Tempo: Key Research Directions and a Consensus Change in Terminology to Cognitive Disengagement Syndrome". Journal of the American Academy of Child and Adolescent Psychiatry. 62 (6): 629–645. doi:10.1016/j.jaac.2022.07.821. PMC 9943858. PMID 36007816.
- ^ Wietecha L, Williams D, Shaywitz S, Shaywitz B, Hooper SR, Wigal SB, et al. (November 2013). "Atomoxetine improved attention in children and adolescents with attention-deficit/hyperactivity disorder and dyslexia in a 16 week, acute, randomized, double-blind trial". Journal of Child and Adolescent Psychopharmacology. 23 (9): 605–613. doi:10.1089/cap.2013.0054. PMC 3842866. PMID 24206099.
- ^ Treuer T, Gau SS, Méndez L, Montgomery W, Monk JA, Altin M, et al. (April 2013). "A systematic review of combination therapy with stimulants and atomoxetine for attention-deficit/hyperactivity disorder, including patient characteristics, treatment strategies, effectiveness, and tolerability". Journal of Child and Adolescent Psychopharmacology. 23 (3): 179–193. doi:10.1089/cap.2012.0093. PMC 3696926. PMID 23560600.
- ^ "Parent's Medication Guide: ADHD". American Psychiatric Association (Guidelines (Tertiary source)). American Psychiatric Association & American Academy of Child and Adolescent Psychiatry (AACAP). June 2013. Archived from the original on 2 February 2017. Retrieved 1 January 2017.
Though not FDA-approved for combined treatment, atomoxetine (Strattera) is sometimes used in conjunction with stimulants as an off-label combination therapy.
- ^ Malenka RC, Nestler EJ, Hyman SE, Holtzman DM (2015). "14: Higher Cognitive Function and Behavioral Control". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (3 ed.). New York: McGraw-Hill Medical. ISBN 9780071827706.
- ^ Chan E, Fogler JM, Hammerness PG (May 2016). "Treatment of Attention-Deficit/Hyperactivity Disorder in Adolescents: A Systematic Review". JAMA. 315 (18): 1997–2008. doi:10.1001/jama.2016.5453. PMID 27163988.
- ^ Sofuoglu M, DeVito EE, Waters AJ, Carroll KM (January 2013). "Cognitive enhancement as a treatment for drug addictions". Neuropharmacology. 64 (1): 452–463. doi:10.1016/j.neuropharm.2012.06.021. PMC 3445733. PMID 22735770.
- ^ Cite error: The named reference
:0was invoked but never defined (see the help page). - ^ Cite error: The named reference
:1was invoked but never defined (see the help page). - ^ Cite error: The named reference
:2was invoked but never defined (see the help page). - ^ Cite error: The named reference
:3was invoked but never defined (see the help page). - ^ British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 344–345. ISBN 9780857113382.
- ^ "Atomoxetine Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 22 March 2019. Retrieved 3 March 2019.
- ^ Cite error: The named reference
Ornoy2018was invoked but never defined (see the help page). - ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Atomoxetine - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.