 Global Information
Global InformationDexlansoprazole information
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Kapidex, Dexilant, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a695020 |
| License data |
|
| Routes of administration | By mouth |
| Drug class | Proton pump inhibitor |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Excretion | 50% renal and 47% in the feces[2] |
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID |
|
| IUPHAR/BPS |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEMBL |
|
| ECHA InfoCard | 100.215.667 |
| Chemical and physical data | |
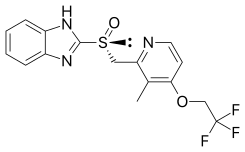
| Formula | C16H14F3N3O2S |
| Molar mass | 369.36 g·mol−1 |
| 3D model (JSmol) |
|
SMILES
| |
InChI
| |
| | |
Dexlansoprazole, is a medication which reduces stomach acid.[3] It is used to treat gastroesophageal reflux disease.[3] Effectiveness is similar to other proton pump inhibitors (PPIs).[4] It is taken by mouth.[3]
Common side effects include diarrhea, abdominal pain, and nausea.[3] Serious side effects may include osteoporosis, low blood magnesium, Clostridium difficile infection, anaphylaxis, and pneumonia.[3] Use in pregnancy and breastfeeding is of unclear safety.[5] It works by blocking H+/K+-ATPase in the parietal cells of the stomach.[3]
Dexlansoprazole was approved for medical use in the United States in 2009.[3] In Canada in 2016, it was the most expensive PPI available.[4] In 2020, it was the 263rd most commonly prescribed medication in the United States, with more than 1 million prescriptions.[6][7]
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ Product Information: DEXILANT delayed release oral capsules, dexlansoprazole delayed release oral capsules. Takeda Pharmaceuticals, Inc., Deerfield, IL, 2010. Revised: September 2012
- ^ a b c d e f g "Dexlansoprazole Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 3 March 2019.
- ^ a b "[99] Comparative effectiveness of proton pump inhibitors | Therapeutics Initiative". 28 June 2016. Retrieved 14 July 2016.
- ^ "Dexlansoprazole Use During Pregnancy". Drugs.com. Retrieved 3 March 2019.
- ^ "The Top 300 of 2020". ClinCalc. Retrieved 7 October 2022.
- ^ "Dexlansoprazole - Drug Usage Statistics". ClinCalc. Retrieved 7 October 2022.