 Global Information
Global InformationDendrimer information

Dendrimers are highly ordered, branched polymeric molecules.[1][2] Synonymous terms for dendrimer include arborols and cascade molecules. Typically, dendrimers are symmetric about the core, and often adopt a spherical three-dimensional morphology. The word dendron is also encountered frequently. A dendron usually contains a single chemically addressable group called the focal point or core. The difference between dendrons and dendrimers is illustrated in the top figure, but the terms are typically encountered interchangeably.[3]
Dendrimer Substance composed of identical dendrimer molecules.
Dendrimer molecule
Molecule consisting of one or more dendrons emanating from a single constitutional unit.
Dendron
Part of a molecule with only one free valence, comprising exclusively dendritic and terminal constitutional repeating units and in which each path from the free valence to any end-group comprises the same number of constitutional repeating units. Note 1: For the purpose of determining the nature of constitutional repeating units the free valence is treated as a connection to a CRU. Note 2: A dendrimer molecule comprising only one dendron is sometimes referred to as dendron, monodendron or functionalised dendron. The use of the terms 'dendron' or 'monodendron' in the meaning of molecule or substance is not acceptable. Note 3: In a dendron, macrocycles of constitutional units are absent.[4]

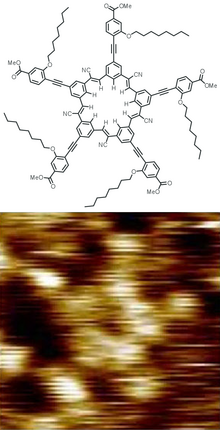
The first dendrimers were made by divergent synthesis approaches by Fritz Vögtle in 1978,[7] R.G. Denkewalter at Allied Corporation in 1981,[8][9] Donald Tomalia at Dow Chemical in 1983[10] and in 1985,[11][12] and by George R. Newkome in 1985.[13] In 1990 a convergent synthetic approach was introduced by Craig Hawker and Jean Fréchet.[14] Dendrimer popularity then greatly increased, resulting in more than 5,000 scientific papers and patents by the year 2005.
- ^ Astruc D, Boisselier E, Ornelas C (April 2010). "Dendrimers designed for functions: from physical, photophysical, and supramolecular properties to applications in sensing, catalysis, molecular electronics, photonics, and nanomedicine". Chemical Reviews. 110 (4): 1857–959. doi:10.1021/cr900327d. PMID 20356105.
- ^ Vögtle, Fritz / Richardt, Gabriele / Werner, Nicole Dendrimer Chemistry Concepts, Syntheses, Properties, Applications 2009 ISBN 3-527-32066-0
- ^ Nanjwade BK, Bechra HM, Derkar GK, Manvi FV, Nanjwade VK (October 2009). "Dendrimers: emerging polymers for drug-delivery systems". European Journal of Pharmaceutical Sciences. 38 (3): 185–96. doi:10.1016/j.ejps.2009.07.008. PMID 19646528.
- ^ Fradet, Alain; Chen, Jiazhong; Hellwich, Karl-Heinz; Horie, Kazuyuki; Kahovec, Jaroslav; Mormann, Werner; Stepto, Robert F. T.; Vohlídal, Jiří; Wilks, Edward S. (2019-03-26). "Nomenclature and terminology for dendrimers with regular dendrons and for hyperbranched polymers (IUPAC Recommendations 2017)". Pure and Applied Chemistry. 91 (3): 523–561. doi:10.1515/pac-2016-1217. ISSN 0033-4545.
- ^ Bauer, Roland. E.; Enkelmann, Volker; Wiesler, Uwe M.; Berresheim, Alexander J.; Müllen, Klaus (2002). "Single-Crystal Structures of Polyphenylene Dendrimers". Chemistry: A European Journal. 8 (17): 3858–3864. doi:10.1002/1521-3765(20020902)8:17<3858::AID-CHEM3858>3.0.CO;2-5. PMID 12203280.
- ^ Hirsch BE, Lee S, Qiao B, Chen CH, McDonald KP, Tait SL, Flood AH (September 2014). "Anion-induced dimerization of 5-fold symmetric cyanostars in 3D crystalline solids and 2D self-assembled crystals". Chemical Communications. 50 (69): 9827–30. doi:10.1039/C4CC03725A. PMID 25080328. S2CID 12439952.
- ^ Buhleier E, Wehner W, Vogtle F (1978). ""Cascade"- and "Nonskid-Chain-like" Syntheses of Molecular Cavity Topologies". Synthesis. 1978 (2): 155–158. doi:10.1055/s-1978-24702.
- ^ U.S. patent 4,289,872 Denkewalter, Robert G., Kolc, Jaroslav, Lukasavage, William J.
- ^ Denkewalter, Robert G. et al. (1981) "Macromolecular highly branched homogeneous compound" U.S. patent 4,410,688
- ^ Tomalia, Donald A. and Dewald, James R. (1983) "Dense star polymers having core, core branches, terminal groups" U.S. patent 4,507,466
- ^ Tomalia DA, Baker H, Dewald J, Hall M, Kallos G, Martin S, Roeck J, Ryder J, Smith P (1985). "A New Class of Polymers: Starburst-Dendritic Macromolecules". Polymer Journal. 17: 117–132. doi:10.1295/polymj.17.117.
- ^ "Treelike molecules branch out – chemist Donald A. Tomalia synthesized first dendrimer molecule – Chemistry – Brief Article". Science News. 1996.
- ^ Newkome GR, Yao Z, Baker GR, Gupta VK (1985). "Micelles. Part 1. Cascade molecules: a new approach to micelles. A [27]-arborol". J. Org. Chem. 50 (11): 2003–2004. doi:10.1021/jo00211a052.
- ^ Hawker CJ, Fréchet JM (1990). "Preparation of polymers with controlled molecular architecture. A new convergent approach to dendritic macromolecules". J. Am. Chem. Soc. 112 (21): 7638–7647. doi:10.1021/ja00177a027.