 Global Information
Global InformationAlendronic acid information
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Fosamax, Binosto, others |
| Other names | Alendronate, alendronate sodium (USAN US) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601011 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 0.6% |
| Metabolism | excreted unchanged |
| Elimination half-life | 126 months |
| Excretion | Kidney |
| Identifiers | |
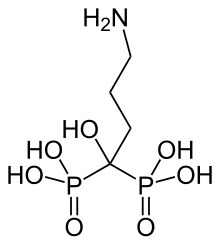
IUPAC name
| |
| CAS Number |
|
| PubChem CID |
|
| IUPHAR/BPS |
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEBI |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) |
|
| ECHA InfoCard | 100.128.415 |
| Chemical and physical data | |
| Formula | C4H13NO7P2 |
| Molar mass | 249.096 g·mol−1 |
| 3D model (JSmol) |
|
SMILES
| |
InChI
| |
| | |
Alendronic acid, sold under the brand name Fosamax among others, is a bisphosphonate medication used to treat osteoporosis and Paget's disease of bone.[3] It is taken by mouth.[3] Use is often recommended together with vitamin D, calcium supplementation, and lifestyle changes.[3]
Common side effects (1 to 10% of patients) include constipation, abdominal pain, nausea, and acid reflux.[3] Use is not recommended during pregnancy or in those with poor kidney function.[4] Alendronic acid works by decreasing the activity of cells that break down bone.[3]
Alendronic acid was first described in 1978 and approved for medical use in the United States in 1995.[3][5] It is available as a generic medication. In 2021, it was the 94th most commonly prescribed medication in the United States, with more than 7 million prescriptions.[6][7]
- ^ "Alendronate Use During Pregnancy". Drugs.com. 22 August 2019. Retrieved 17 May 2020.
- ^ "Product monograph brand safety updates". Health Canada. 7 July 2016. Retrieved 3 April 2024.
- ^ a b c d e f "Fosamax Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 2 February 2019.
- ^ British National Formulary : BNF 76 (76 ed.). Pharmaceutical Pres s. 2018. pp. 710–711. ISBN 978-0-85711-338-2.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 523. ISBN 978-3-527-60749-5.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Alendronate - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.