 Global Information
Global InformationLorlatinib information
 | |
| Clinical data | |
|---|---|
| Trade names | Lorbrena, Lorviqua |
| Other names | PF-6463922 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a619005 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 81% |
| Protein binding | 66% |
| Metabolism | Mainly CYP3A4 and UGT1A4 |
| Elimination half-life | 24 hrs (single dose) |
| Excretion | 48% urine (<1% unchanged), 41% faeces (9% unchanged) |
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID |
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEBI |
|
| CompTox Dashboard (EPA) |
|
| ECHA InfoCard | 100.245.079 |
| Chemical and physical data | |
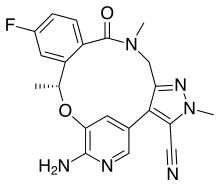
| Formula | C21H19FN6O2 |
| Molar mass | 406.421 g·mol−1 |
| 3D model (JSmol) |
|
SMILES
| |
InChI
| |
Lorlatinib, sold under the brand name Lorbrena in the United States, Canada, and Japan, and Lorviqua in the European Union, is an anti-cancer medication used for the treatment of non-small cell lung cancer.[4] It is an orally administered inhibitor of anaplastic lymphoma kinase (ALK) and C-ros oncogene 1 (ROS1), two enzymes that play a role in the development of cancer.[6] It was developed by Pfizer.[7]
The most common adverse reactions include edema, peripheral neuropathy, weight gain, cognitive effects, fatigue, dyspnea, arthralgia, diarrhea, mood effects, hypercholesterolemia, hypertriglyceridemia, and cough.[7]
Lorlatinib was approved for medical use in the United States in November 2018,[8][9] and in the European Union in May 2019.[5]
- ^ a b "Lorviqua APMDS". Therapeutic Goods Administration (TGA). 26 May 2022. Archived from the original on 10 March 2024. Retrieved 10 March 2024.
- ^ "Lorbrena Product information". Health Canada. Archived from the original on 30 May 2022. Retrieved 29 May 2022.
- ^ "Summary Basis of Decision (SBD) for Lorbrena". Health Canada. 23 October 2014. Archived from the original on 31 May 2022. Retrieved 29 May 2022.
- ^ a b "Lorbrena- lorlatinib tablet, film coated". DailyMed. 7 September 2023. Archived from the original on 7 May 2024. Retrieved 4 June 2024.
- ^ a b "Lorviqua EPAR". European Medicines Agency (EMA). 6 May 2019. Archived from the original on 14 November 2021. Retrieved 4 June 2024.
- ^ Cite error: The named reference
Nagasaka_2020was invoked but never defined (see the help page). - ^ a b Cite error: The named reference
FDA 20210304was invoked but never defined (see the help page). - ^ Cite error: The named reference
FDAwas invoked but never defined (see the help page). - ^ "Drug Approval Package: Lorbrena (lorlatinib)". U.S. Food and Drug Administration (FDA). 10 December 2018. Archived from the original on 8 November 2022. Retrieved 4 June 2024.