 Global Information
Global InformationHafnium trifluoromethanesulfonate information
| |
| Names | |
|---|---|
| IUPAC names
Hafnium(IV) trifluoromethanesulfonate
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider |
|
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
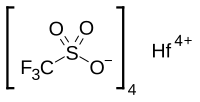
Chemical formula
|
Hf(OTf)4 |
| Molar mass | 774.8 g/mol |
| Appearance | Colourless solid |
| Melting point | 350 °C (662 °F; 623 K) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
irritantant |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H314, H315, H319, H335 |
Precautionary statements
|
P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 |
| Flash point | Non-flammable |
| Safety data sheet (SDS) | [1] |
| Related compounds | |
Other anions
|
Hafnium tetrachloride Hafnium tetrafluoride Hafnium(IV) bromide Hafnium(IV) iodide |
Other cations
|
Titanium(IV) triflate Zirconium(IV) triflate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Hafnium(IV) triflate or hafnium trifluoromethansulfonate is a salt with the formula Hf(OSO2CF3)4, also written as Hf(OTf)4. Hafnium triflate is used as an impure mixture as a catalyst. Hafnium (IV) has an ionic radius of intermediate range (Al < Ti < Hf < Zr < Sc < Ln) and has an oxophilic hard character typical of group IV metals. This solid is a stronger Lewis acid than its typical precursor hafnium tetrachloride, HfCl4, because of the strong electron-withdrawing nature of the four triflate groups, which makes it a great Lewis acid and has many uses including as a great catalyst at low Lewis acid loadings for electrophilic aromatic substitution and nucleophilic substitution reactions.[1]
- ^ Ishitani, Haruro; Suzuki, Hirotsugu; Saito, Yuki; Yamashita, Yasuhiro; Kobayashi, Shū (2015). "Hafnium Trifluoromethanesulfonate [Hf(OTf)4] as a Unique Lewis Acid in Organic Synthesis". European Journal of Organic Chemistry. 2015 (25): 5485–5499. doi:10.1002/ejoc.201500423. ISSN 1099-0690.