 Global Information
Global InformationFlow battery information
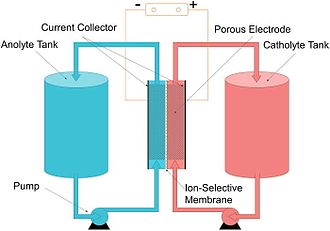
A flow battery, or redox flow battery (after reduction–oxidation), is a type of electrochemical cell where chemical energy is provided by two chemical components dissolved in liquids that are pumped through the system on separate sides and in opposite direction of a membrane.[2][3] Ion transfer inside the cell (accompanied by flow of electric current through an external circuit) occurs through the membrane while both liquids circulate in their own respective space. Cell voltage is chemically determined by the Nernst equation and ranges, in practical applications, from 1.0 to 2.43 volts. The energy capacity is a function of the electrolyte volume and the power is a function of the surface area of the electrodes.[4]
Various types of flow batteries have been demonstrated, including inorganic flow batteries[5] and organic flow batteries.[6] Under each category, flow battery design can be further classified into full flow batteries, semi-flow batteries, and membraneless flow batteries. The fundamental difference between conventional and flow batteries is that energy is stored in the electrode material in conventional batteries, while in flow batteries it is stored in the electrolyte. Patent Classifications for Flow Batteries have not been fully developed as of 2021. Cooperative Patent Classification considers RFBs as a subclass of regenerative fuel cell (H01M8/18), even though it is more appropriate to consider fuel cells as a subclass of flow batteries.
A flow battery may be used like a fuel cell (where new charged negolyte (a.k.a. reducer or fuel) and charged posolyte (a.k.a. oxidant) are added to the system) or like a rechargeable battery (where an electric power source drives regeneration of the reducer and oxidant). While flow batteries have certain technical advantages over conventional rechargeable batteries with solid electroactive materials, such as independent scaling of power (determined by the size of the stack) and of energy (determined by the size of the tanks), long cycle and calendar life,[7] and potentially lower total cost of ownership, all flow batteries suffer from low cycle energy efficiency (50–80%). This inferior energy efficiency stems from the need to operate flow batteries at high (>= 100 mA/cm2) current densities to reduce the effect of the internal crossover (through the membrane/separator inside each cell) and to reduce the cost of power (size of stacks). Also, most flow batteries (Zn-Cl2, Zn-Br2 and H2-LiBrO3 are exceptions) have lower specific energy (heavier weight) compared to lithium-ion batteries. The heavier weight results mostly from the need to use a solvent (usually-water) to keep the redox active species in the liquid phase.[8]
- ^ Qi, Zhaoxiang; Koenig, Gary M. (12 May 2017). "Review Article: Flow battery systems with solid electroactive materials". Journal of Vacuum Science & Technology B, Nanotechnology and Microelectronics: Materials, Processing, Measurement, and Phenomena. 35 (4): 040801. Bibcode:2017JVSTB..35d0801Q. doi:10.1116/1.4983210. ISSN 2166-2746.
- ^ Badwal, Sukhvinder P. S.; Giddey, Sarbjit S.; Munnings, Christopher; Bhatt, Anand I.; Hollenkamp, Anthony F. (24 September 2014). "Emerging electrochemical energy conversion and storage technologies". Frontiers in Chemistry. 2: 79. Bibcode:2014FrCh....2...79B. doi:10.3389/fchem.2014.00079. PMC 4174133. PMID 25309898.
- ^ Alotto, P.; Guarnieri, M.; Moro, F. (2014). "Redox Flow Batteries for the storage of renewable energy: a review". Renewable & Sustainable Energy Reviews. 29: 325–335. doi:10.1016/j.rser.2013.08.001. hdl:11577/2682306.
- ^ Yuriy V. Tolmachev "Review—Flow Batteries from 1879 to 2022 and Beyond." 2023 J. Electrochem. Soc. 170 030505. https://iopscience.iop.org/article/10.1149/1945-7111/acb8de/meta
- ^ Hu, B.; Luo, J.; DeBruler C.; Hu, M; Wu, W.; Liu, T. L. (2019). Redox Active Inorganic Materials for Redox Flow Batteries in Encyclopedia of Inorganic and Bioinorganic Chemistry: Inorganic Battery Materials. pp. 1–25.
- ^ Luo, J.; Hu, B.; Hu, M.; Liu, T. L. (13 September 2019). "Status and Prospects of Organic Redox Flow Batteries towards Renewable Energy Storage". ACS Energy Lett. 2019, 4 (9): 2220–2240. doi:10.1021/acsenergylett.9b01332. S2CID 202210484.
- ^ Clark, Elliot (17 November 2023). "What is a Calendar-life?". Energy Theory. Retrieved 3 May 2024.
- ^ Yuriy V. Tolmachev; Svetlana V. Starodubceva (2022). "Flow batteries with solid energy boosters". Electrochemical Science and Engineering. 12 (4): 731–766. doi:10.5599/jese.1363.