 Global Information
Global InformationTungsten oxytetrafluoride information
| |
| Identifiers | |
|---|---|
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
WOF4 |
| Molar mass | 275.83 g/mol |
| Appearance | colourless crystals[1] |
| Density | 5.07 g/cm3[2] |
| Melting point | 110[2] °C (230 °F; 383 K) |
| Boiling point | 185[2] °C (365 °F; 458 K) |
Solubility in water
|
reacts[2] |
| Solubility | soluble in chloroform[3] sparingly soluble in carbon disulfide[3] |
| Structure | |
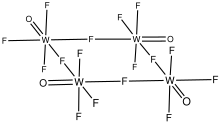
Crystal structure
|
monoclinic |
| Related compounds | |
Other anions
|
Tungsten(VI) oxytetrachloride Tungsten(VI) oxytetrabromide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Tungsten oxytetrafluoride is an inorganic compound with the formula WOF4. It is a colorless diamagnetic solid. The compound is one of many oxides of tungsten. It is usually encountered as product of the partial hydrolysis of tungsten hexafluoride.
- ^ Perry, Dale L.; Phillips, Sidney L. (1995). Handbook of inorganic compounds. Boca Raton: CRC Press. p. 428. ISBN 0-8493-8671-3. OCLC 32347397.
- ^ a b c d Haynes, William M.; Lide, David R.; Bruno, Thomas J. (2017). CRC handbook of chemistry and physics : a ready-reference book of chemical and physical data. Boca Raton, Florida. p. 104. ISBN 978-1-4987-5429-3. OCLC 957751024.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ a b Cite error: The named reference
Erik Lassner, Wolf-Dieter Schubertwas invoked but never defined (see the help page).