 Global Information
Global InformationSodium monothiophosphate information
| |
| Names | |
|---|---|
| IUPAC name
Sodium monothiophosphate
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ECHA InfoCard | 100.030.224 |
| EC Number |
|
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
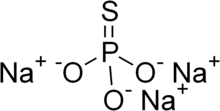
Chemical formula
|
Na3PO3S |
| Molar mass | 180.00 g·mol−1 |
| Appearance | White solid |
| Density | 1.58 g/cm3 (dodecahydrate) 2.40 g/cm3 (anhydrous) |
| Melting point | 120 to 125 °C (248 to 257 °F; 393 to 398 K) (decomposition) |
Solubility in water
|
soluble |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Sodium monothiophosphate, or sodium phosphorothioate, is an inorganic compound with the chemical formula Na3PO3S. It is a sodium salt of monothiophosphoric acid (H3PO3S). Sodium monothiophosphate forms hydrates Na3PO3S·xH2O. The anhydrous form and all hydrates are white solids. The anhydrous salt (x = 0) (Na3PO3S) decomposes without melting at 120-125 °C. More common is the dodecahydrate (Na3PO3S·12H2O). A nonahydrate is also known (Na3PO3S·9H2O).
Related salts are the sodium dithiophosphate undecahydrate Na3PO2S2·11H2O, sodium trithiophosphate undecahydrate Na3POS3·11H2O, and sodium tetrathiophosphate octahydrate Na3PS4·8H2O.[1]
- ^ Elias, D. P. (1957). "Crystallographic Data on Some Sodium Phosphorothioates". Acta Crystallographica. 10 (9): 600. Bibcode:1957AcCry..10..600E. doi:10.1107/S0365110X57002108.