 Global Information
Global InformationMusk xylene information
| |
| Names | |
|---|---|
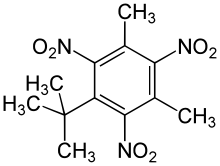
| Preferred IUPAC name
1-tert-Butyl-3,5-dimethyl-2,4,6-trinitrobenzene | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI |
|
| ChEMBL |
|
| ChemSpider |
|
| ECHA InfoCard | 100.001.210 |
| EC Number |
|
| KEGG |
|
| MeSH | musk+xylene |
PubChem CID
|
|
| UNII |
|
| UN number | 2956 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C 12H 15N 3O 6 |
| Molar mass | 297.2640 g mol−1 |
| Appearance | Yellow crystals |
| Odor | Musk-like |
| Melting point | 110 °C (230 °F; 383 K) |
Solubility in water
|
150 ng dm−1 |
| log P | 4.369 |
| Vapor pressure | 9.7 mPa (at 40 °C) |
| Hazards | |
| GHS labelling: | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H201, H351, H410 |
Precautionary statements
|
P201, P202, P210, P230, P240, P250, P273, P280, P281, P308+P313, P370+P380, P372, P373, P391, P401, P405, P501 |
| Flash point | 2 °C (36 °F; 275 K) |
Autoignition
temperature |
305 to 341 °C (581 to 646 °F; 578 to 614 K) |
| Related compounds | |
Related nitro musks
|
Musk ambrette |
Related compounds
|
Trinitrotoluene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Musk xylene is a synthetic musk fragrance which mimics natural musk. It has been used as a perfume fixative in a wide variety of consumer products, and is still used in some cosmetics and fragrances.
Musk xylene was once the most widely used of the "nitro-musks", but its use has declined sharply since the mid-1980s due to safety and environmental concerns. Its explosive and carcinogenic hazards are recognized to be borderline, and musk xylene is a useful example of the lowest level of such risks which need to be taken into account. However, it is a very persistent and very bioaccumulative pollutant in the aquatic environment (vPvB substance), and is the first substance to be proposed as a "substance of very high concern" (SVHC) for these reasons alone under the European Union REACH Regulation. Since no company has applied for authorisation, it is banned in the EU.[2]
- ^ Section 1.3, European Union Risk Assessment Report (2005), pp. 6–7.
- ^ "Are there safer alternatives? - ECHA". Archived from the original on 2015-06-16. Retrieved 2015-05-18.