 Global Information
Global InformationIsavuconazonium information
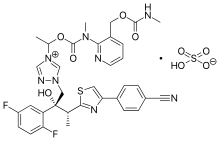 | |
| Clinical data | |
|---|---|
| Trade names | Cresemba |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous (IV) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| CompTox Dashboard (EPA) |
|
| Chemical and physical data | |
| Formula | C35H35F2N8O5S |
| Molar mass | 717.77 g·mol−1 |
| 3D model (JSmol) |
|
SMILES
| |
InChI
| |
| | |
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Cresemba |
| Other names | BAL8557 |
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous (IV) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEBI |
|
| ChEMBL |
|
| NIAID ChemDB |
|
| CompTox Dashboard (EPA) |
|
| Chemical and physical data | |
| Formula | C22H17F2N5OS |
| Molar mass | 437.47 g·mol−1 |
| 3D model (JSmol) |
|
| Melting point | 122[11] °C (252 °F) |
| Solubility in water | 14.2 ± 0.5 × 10−6 mol/L (pH 7.4)[11] |
SMILES
| |
InChI
| |
| | |
Isavuconazonium sulfate, sold under the brand name Cresemba, is a systemic antifungal medication of the triazole class which is used to treat invasive aspergillosis and mucormycosis.[9][7][8][12]
The most common side effects include abnormal liver tests, nausea, vomiting, difficulty breathing, abdominal pain, diarrhea, injection site reactions, headache, low blood potassium and skin rash.[10]
Isavuconazonium is a prodrug of isavuconazole.[13]
- ^ a b "Isavuconazonium (Cresemba) Use During Pregnancy". Drugs.com. 31 January 2020. Retrieved 26 August 2020.
- ^ "Cresemba isavuconazole (as isavuconazonium sulfate) 200 mg powder for injection vial". Therapeutic Goods Administration (TGA). ARTG ID 305480. Archived from the original on 19 June 2022. Retrieved 5 September 2021.
- ^ "Cresemba isavuconazole (as isavuconazonium sulfate) 100 mg capsule blister pack". Therapeutic Goods Administration (TGA). ARTG ID 305452. Archived from the original on 5 September 2021. Retrieved 5 September 2021.
- ^ "AusPAR: Isavuconazole (as sulphate)". Therapeutic Goods Administration (TGA). 22 January 2020. Retrieved 5 September 2021.
- ^ "Regulatory Decision Summary for Cresemba". Drug and Health Product Register. 23 October 2014.
- ^ "Drug and medical device highlights 2018: Helping you maintain and improve your health". Health Canada. 14 October 2020. Retrieved 17 April 2024.
- ^ a b "Cresemba 100 mg hard capsules - Summary of Product Characteristics (SmPC)". (emc). 28 June 2021. Retrieved 5 September 2021.
- ^ a b "Cresemba 200mg Powder for concentrate for solution for infusion - Summary of Product Characteristics (SmPC)". (emc). 28 June 2021. Retrieved 5 September 2021.
- ^ a b "Cresemba- isavuconazonium sulfate capsule Cresemba- isavuconazonium sulfate injection, powder, lyophilized, for solution". DailyMed. 2 December 2019. Retrieved 26 August 2020.
- ^ a b Cite error: The named reference
Cresemba EPARwas invoked but never defined (see the help page). - ^ a b Cite error: The named reference
Voronin_2021was invoked but never defined (see the help page). - ^ Donnelley MA, Zhu ES, Thompson GR (2 June 2016). "Isavuconazole in the treatment of invasive aspergillosis and mucormycosis infections". Infection and Drug Resistance. 9: 79–86. doi:10.2147/IDR.S81416. PMC 4898026. PMID 27330318.
- ^ Wilkes GM, Barton-Burke M (2019). 2020-2021 Oncology Nursing Drug Handbook. Jones & Bartlett Learning. pp. 1874–1876. ISBN 978-1-284-17132-7.