 Global Information
Global InformationHinokinin information
This article may need to be rewritten to comply with Wikipedia's quality standards. (March 2020) |
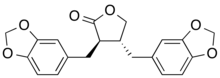
| |
| Names | |
|---|---|
| Preferred IUPAC name
(3R,4R)-3,4-Bis[(2H-1,3-benzodioxol-5-yl)methyl]oxolan-2-one | |
| Other names
(3R,4R)-3,4-Bis(1,3-benzodioxol-5-ylmethyl)dihydro-2(3H)-furanone
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL |
|
| ChemSpider |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C20H18O6 |
| Molar mass | 354.358 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Hinokinin is a dibenzylbutyrolactone lignan, derived from various species of plants. It is a potential antichagonistic agent.[1] In vitro, it has been shown to have potential neuroprotective effects[2] as well as anti-inflammatory, anti-tumor, antiviral and antifungal properties.[3]
Hinokinin was isolated for the first time by Yoshiki and Ishiguro in 1933 from hinoki wood.[4]
- ^ Marcotullio, Maria; Pelosi, Azzurra; Curini, Massimo (2014-09-17). "Hinokinin, an Emerging Bioactive Lignan". Molecules. 19 (9): 14862–14878. doi:10.3390/molecules190914862. ISSN 1420-3049. PMC 6271885. PMID 25232707.
- ^ Timple, Julie Marie V.; Magalhães, Lizandra Guidi; Souza Rezende, Karen Cristina; Pereira, Ana Carolina; Cunha, Wilson Roberto; Andrade e Silva, Márcio Luis; Mortensen, Ole Valente; Fontana, Andréia C. K. (2013-10-10). "The Lignan (−)-Hinokinin Displays Modulatory Effects on Human Monoamine and GABA Transporter Activities". Journal of Natural Products. 76 (10): 1889–1895. doi:10.1021/np400452n. ISSN 0163-3864. PMID 24112084.
- ^ Zhou, Qi-Long; Wang, Hui-Jing; Tang, Pei; Song, Hao; Qin, Yong (October 2015). "Total Synthesis of Lignan Lactone (–)-Hinokinin". Natural Products and Bioprospecting. 5 (5): 255–261. doi:10.1007/s13659-015-0073-3. ISSN 2192-2195. PMC 4607678. PMID 26458924.
- ^ Yoshiki, Y.; Ishiguro, T. (1933). "Ueber die kristallisierten Bestandteile des Hinokiöls". Yakugaku Zasshi. 53 (2): 73–151. doi:10.1248/yakushi1881.53.2_73. ISSN 0031-6903.