 Global Information
Global InformationHexaamminenickel chloride information
| |

| |
| Names | |
|---|---|
| IUPAC name
Hexaamminenickel(II) chloride
| |
| Other names
Nickel hexammine chloride, hexamminenickel chloride
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider |
|
| ECHA InfoCard | 100.149.740 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Cl2H18N6Ni |
| Molar mass | 231.78 g·mol−1 |
| Appearance | violet solid |
| Density | 1.51 g/cm3 |
| Melting point | decomposes |
| Solubility | soluble in NH3 |
| Structure | |
Coordination geometry
|
octahedral |
Dipole moment
|
0 D |
| Related compounds | |
Other cations
|
[Cr(NH3)6]Cl3 [Co(NH3)6]Cl3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
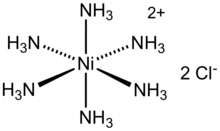
Hexaamminenickel chloride is the chemical compound with the formula [Ni(NH3)6]Cl2. It is the chloride salt of the metal ammine complex [Ni(NH3)6]2+. The cation features six ammonia (called ammines in coordination chemistry) ligands attached to the nickel(II) ion.[1]
- ^ Eßmann, Ralf; Kreiner, Guido; Niemann, Anke; Rechenbach, Dirk; Schmieding, Axel; Sichla, Thomas; Zachwieja, Uwe; Jacobs, Herbert (1996). "Isotype Strukturen einiger Hexaamminmetall(II)‐halogenide von 3d‐Metallen: [V(NH3)6]I2, [Cr(NH3)6]I2, [Mn(NH3)6]Cl2, [Fe(NH3)6]Cl2, [Fe(NH3)6]Br2, [Co(NH3)6]Br2, und [Ni(NH3)6]Cl2". Zeitschrift für anorganische und allgemeine Chemie. 622 (7): 1161–1166. doi:10.1002/zaac.19966220709.