 Global Information
Global InformationCyameluric acid information
 Trihydroxy (imidic acid) tautomer
| |
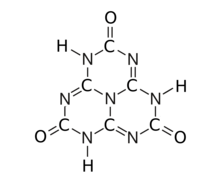 Tricarbonyl (amide) tautomer
| |
| Names | |
|---|---|
| IUPAC name
2,4,6,8,10,12,13-heptazatricyclo[7.3.1.05,13]trideca-1,4,8-triene-3,7,11-trione
| |
| Other names
1,3,4,6,7,9,9b-Heptaazaphenalene-2,5,8(1H,3H,6H)-trione; 2,5,8-trihydroxy-s-heptazine; 1,4,7-trihydro-2,5,8-trioxo-s-heptazine
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
34555, 542266 |
| ChEBI |
|
| ChemSpider |
|
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H3N7O3 |
| Molar mass | 221.13 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Cyameluric acid or 2,5,8-trihydroxy-s-heptazine is a chemical compound with formula C
6N
7O
3H
3, usually described as a heptazine molecule with the hydrogen atoms replaced by hydroxyl groups –OH; or any of its tautomers.
The substance exists as an equilibrium of 17 tautomers that easily interconvert among each other. Calculations show that the symmetric tri-oxo form (1,4,7-trihydro-2,5,8-trioxo-s-heptazine) is the most stable.[1] Therefore, this compound contains amide groups rather than imidic acids.
- ^ Cite error: The named reference
alko2004was invoked but never defined (see the help page).